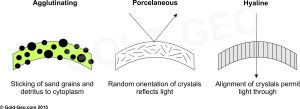
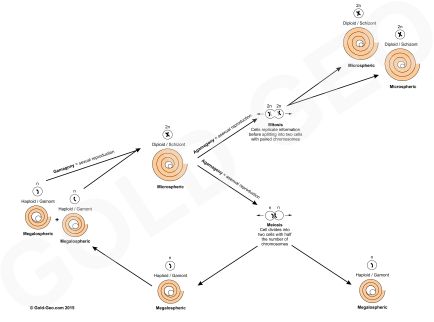
The Cenozoic was an important time of global tectonic reorganisation; India was colliding with Asia, there was no land between North and South America creating a link between the Atlantic and Pacific oceans, the Andes mountain range was rising, Africa was colliding with Europe, and Australia with Southeast Asia. All these events created distinct global bioprovinces of flora and fauna, including larger benthic foraminifera.
There are three main global bioprovinces; the American, Tethyan and Indo-Pacific provinces. Each bioprovince has its own unique evolutionary history including speciation and extinction events, and migratory events from other provinces (BouDagher-Fadel, 2018). It is because each province is unique that global temporal range charts of, especially larger benthic, foraminifera are not reliable. Regional biostratigraphy and biogeography of particular provinces should be studied.
During the Neogene global atmospheric and ocean circulation began to settle in to their present day arrangement. However, at the start of the Aquitanian the Tethyan seaway remained open enabling the migration of foraminifera between the proto-Mediterranean Sea and proto-Indian Ocean. During this time lineages such as the miogypsinids and lepidocyclinids that originated in the Americas had migrated into the Tethyan bioprovince by reaching North Africa on ocean currents and debris. Cycloclypeus speciated in the Tethyan province and migrated quickly east towards Southeast Asia but not west towards the Americas. At the end of the Aquitanian, global extinctions were low and only 8% of global larger benthic foraminifera (LBF) became extinct. The Indo-Pacific bioprovince, however, was an exception to this rule and of the global 8%, 43% of the LBF in the region became extinct opening up new ecological niches to be exploited (BouDagher-Fadel, 2018).
The Burdigalian saw much diversification of species of LBF and new genera of Rotalioids within the Tethyan province, but few in the Indo-Pacific. Acervulinids and planorbulinids were common globally, soritids became diverse and nummulitids and lepidocyclinids thrived together in reef and fore-reef environments. Lepidocyclinids and miogypsinids continued their migration from the Americas to the Indo-Pacific via the Tethys by algal rafting and planktonic gametes through the Mediterranean and Arabian coast before spreading fast on ocean currents to Southeast Asia to fill the niches left behind at the end of the Aquitanian (BouDagher-Fadel, 2018).
The end of the Burdigalian saw marked extinction event in which ~30% of global LBFs became extinct. It was most pronounced in the Americas and Tethys where 50% and 40% became extinct respectively. Casualties of this event included the extinction of lepidocyclinids and miogypsinids in the American Province and Cycloclypeus in the Tethys. In the American province this extinction is attributed the eruption of the Colombia River Basalts which may have increased atmospheric, and as a result ocean, CO2 leading to ocean acidification. The extinction of the lepidocyclinids and miogypsinids here also coincides with the extinction of many hermatypic corals. The extinct corals and lepidocyclinids/miogypsinids of the Caribbean and West Atlantic hold on until later in the Indo-Pacific region (BouDagher-Fadel, 2018).
The collision of Africa with Europe in the Serravallian sealed off the eastern proto-Mediterranean Sea and proto-Indian Ocean leading to 60% of global extinctions of LBF at this time occurring in the Tethyan province due to increased salinities (BouDagher-Fadel, 2018). In the Indo-Pacific the lepidocyclinids and miogypsinids finally become extinct at the end of the Serravallian. Cyclocylpeus, which had disappeared from the Mediterranean in the Burdigalian, continued to thrive, and does so to the present day, branching off to form Katacycloclypeus although this form was short-lived with Katacycloclypeus becoming extinct in the middle of the Serravallian.
In the Mid-Late Miocene the partial emergence of the Panamanian Isthmus closed off equatorial Atlantic-Pacific throughflow. This led to a diversification of the Soritoidea in the American province, such as Archais in the Caribbean. Although in the Indo-Pacific, the Soritoidea were dominated by just two genera; Amphisorus and Marginopora.
By the Late Miocene approximately 75% of LBF in the Mediterranean had become extinct due to the Messinian Salinity Crisis (BouDagher-Fadel, 2018). LBF endemic to the Indo-Pacific were not particularly affected and although no new forms emerged, there was a faunal turnover following the extinction of lepidocyclinids and miogypsinids to being dominated by Cycloclypeus, Marginopora, Alveolinella and several other foraminifera.
During the Pliocene, the emergence of new LBFs was absent globally except in the Indo-Pacific which was the only bioprovince in which new species appeared (BouDagher-Fadel, 2018). These were restricted to the western and central tropical Pacific and included the calcarinids such as Calcarina spengleri and Quasirotalia sp., which have preferences towards shallow-water, high-energy environments.
Global climate cooling from the Pliocene to the present day suppressed speciation and diversification so that long-lived and already established foraminifera continued to thrive and adapt. Today, Indo-Pacific sandy shoals are dominated by Calcarina, Baculogypsina, Amphistegina and the foreslopes by Heterostegina, Operculina and Cycloclypeus.
As previously laid out, the global history of foraminiferal migrations and extinctions varies between bioprovinces so that the ‘Indo-Pacific Letter Stages’ (Fig. 1) based purely on LBF were proposed as an alternative to European biostratigraphic stages that were based on mixed molluscan and foraminiferal assemblages. It was noted that molluscan faunas of the Indo-Pacific developed separately from those in Europe so that European stages could not be correlated with confidence to those in Indonesia. In Indonesia foraminifera are more abundant than molluscs and knowledge and recognition of only a few key taxa enabled quick dating of rocks in the field (BouDagher-Fadel, 2018). Therefore the Indo-Pacific Letter Stage (Fig. 1) subdivision of the Cenozoic was first proposed by van der Vlerk and Umbgrove in 1927 and was later refined by Leupold and Van der Vlerk (1931); Adams (1970); Chaproniere (1984); BouDagher-Fadel and Banner (1999). These stages are assemblage zones based on the first and last appearance of key taxa that form a sequence from ‘Ta’ (oldest) to ‘Th’ (youngest; Fig. 1).
The Indo-Pacific Letter Stages are now well-established and their use is widespread. However, they are problematic. Correlation with global chrono- and biostratigraphy is still uncertain, although attempts have been made (Fig. 1). Problems arise from LBF assemblages being strongly facies controlled determined by changes in relative sea-level affecting photic zone, wave base and sediment supply (BouDagher-Fadel, 2018).

References:
Adams, C.G. 1970. A reconsideration of the East Indian letter classification of the Tertiary, Bulletin of the British Museum (Natural History). Geology, 19, 87-137
Boudaugher-Fadel, M.K., 2018. Evolution and geological significance of larger benthic foraminifera. UCL Press.
BouDagher-Fadel, M.K. and Banner, F.T. 1999. Revision of the stratigraphic significance of the Oligocene-Miocene ‘Letter Stages’. Revue de Micropaléontologie, 42, 93-97
Chaproniere, G.C.H. 1984.The Neogene larger foraminiferal sequence in the Australian and New Zealand regions, and its relevance to the East Indies letter stage classification. Palaeogeography, Palaeoclimatlogy, Palaeoecology, 46, 25-35
Leupold, W. and van der Vlerk, I.M. 1931. The Tertiary. Leidsche Geologische Mededelingen, 5, 611-648
van der Vlerk, I.M. and Umbgrove, J.H. 1927. Tertiary Gidsforaminifera van Nederlandisch Oost-Indie. Wetenschappelijke Mededelingen van de Dienst van de Mijnbouw in Nederlandsch-Oost Indie, 6, 3-35