If you were to think about single-celled organisms, you might imagine that they could only been seen under the microscope. Foraminifera (colloquially called ‘forams’), however, are single-celled organisms that can grow from several millimetres up to the size of small frisbees. The one in the above picture, Assilina exponens (Eocene, Germany), is the same size and shape as a ready salted crisp. The largest recorded foram measured 19cm in diameter, nearly 4 times the size of the Assilina exponens pictured above.
Forams are major rock-forming organisms that contribute 43 million tons of carbonate in the oceans per year (Langer et al., 1997) and make up approximately 20% of the carbonate in the geological record. They are also major components of beach ‘sand’ especially in subtropical environments (you may have seen pictures of the beautiful variety of morphologies in foraminifera that make up beach sand that occasionally do the rounds on social media), even the pyramids of Egypt are built from Nummulitic limestone, like the one below, formed of millions of ‘tests’ of foraminifera from the genus Nummulites.


These organisms are important predators and prey in the ocean ecosystem, and make up much of the biomass of the seafloor and water column, it is even estimated that most of the seafloor is even covered by the reticulate pseudopodia of these organisms. Yet, these creatures remain almost unknown to most outside of the geological or biological communities and have yet to achieve their deserved star status having been mostly overlooked on prime-time nature documentaries. So what exactly are they? Why are they so large? And why do people like me spend all day looking at them?
Biology
The taxonomy of foraminifera is problematic and highly disputed. For now, foraminifera are placed within the Rhizaria a ‘supergroup’ of Protists, one of the ‘5 Kingdoms’, although this kingdom is currently being phased out. What is not disputed is that they are eukaryotic single-celled organisms that resemble amoebae with pseudopodia (stringy, gelatinous appendages that are used for locomotion and collecting food).
Foraminifera first appeared during the time of the ‘Cambrian Explosion’ ~542 Ma, with oldest known foraminifera being Platysolenites sp. To date there are approximately 275,000 recognised species, of these 10,000 are extant with remainder extinct and consigned to the fossil record.
There are many ways to divide forams based on their skeletal structure or mode of life. Foraminifera are often described as being ‘benthic’ – living close to the sediment-water interface, or ‘planktonic’ – living within the water column. Planktonic foraminifera account for only around 50 species of 10,000 species around today. Benthic foraminifera account for the remaining extant species, these are often further subdivided by their size into smaller and larger benthic forams, or according to their test structure.
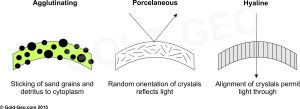
Foraminifera secrete a shell (generally composed of calcite), which is technically called a ‘test’ because cytoplasm covers the exterior of the shell in a ‘sheath’ of ectoplasm. Foraminiferal tests may be ‘agglutinated’ where they are made from sand grains, shell fragments from other organisms, or generally whatever other detritus the foram can find within the substrate in which it lives that it can stick to itself. Foraminiferal tests may be ‘granular/porcelaneous’ such as those of miliolids that do not let light though, or ‘hyaline/glassy’ in which the orientation of crystals permits light to pass giving a transparent or translucent appearance to the test (see below). Again, foraminifera can be further subdivided and subdivided further still based on the numerous shapes, chambers, and apertural openings etc. of the test.
Many forams are symbiotic and grow chambered tests in which to house their symbionts, it is these that make up the larger benthic foraminifera. As their name suggests it is this group that attain the largest sizes. The first formed chamber of a larger benthic foram is called the ‘proloculus’, it is here that the foram keeps its important cellular material with cytoplasm filling the rest of the test. From the proloculus the foram produces a series of large chambers (sometimes divided into smaller ‘chamberlets’). The cytoplasm in the chambers is linked to the proloculus by a canal-like structure called a ‘foramen’ (from which the foraminifera derive their name). It takes forams such as Heterostegina, Elphidium, and Rotalia 24 hours to grow a new chamber. It then takes them a further 5-7 days to recuperate and muster the strength to add a new chamber (Myers, 1941). Within these chambers the larger benthic foraminifera ‘farm’ their symbionts, often photosynthetic cyanobacteria. Rather counter-intuitively the largest larger benthic foraminifera are found within the most oligotrophic (low nutrient) environments. This is because they must reach a large size in order to house the necessary number of cyanobacteria to keep them alive.
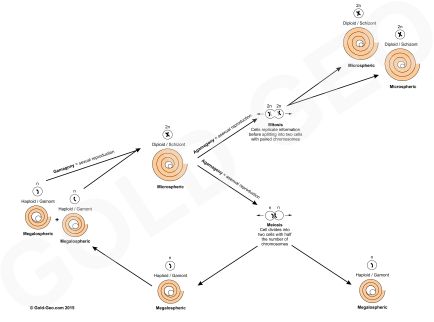
Larger benthic foraminifera live typically from several weeks to a couple of years. Their life span is in part dependent on their reproductive cycle. Foraminifera have a complex life cycle in which they can reproduce sexually or asexually. Asexual reproduction produces a haploid ‘Megalospheric’ morphology via meiosis that contains a single set of chromosomes. Sexual reproduction produces a diploid ‘Microspheric’ morphology that contains a pair of chromosomes (see reproductive cycle figure below). Again rather confusingly, the Megalospheric form is often smaller than the Microspheric form. The terms ‘mega’ and ‘micro’ in this case does not refer to the size of the test but of the proloculus. The Megalospheric morphology has a larger proloculus than the Microspheric morphology.
Uses
Foraminifera are useful for palaeontologists and geologists like me in that they provide environmental information about the rocks in which they are contained were deposited. They are also key index fossils in helping to determine at what time these rocks were laid down. As mentioned previously, many larger benthic forams contain symbiotic cyanobacteria that photosynthesise to survive. Photosynthesis requires sunlight and because of this larger benthic forams are often found within the ‘photic zone’, the zone at which sunlight can still penetrate the water column.
Some smaller benthic foraminifera such as Amphistegina house photosynthetic symbionts adapted to blue light that can penetrate greater water depths of up to 150m, although healthy populations of Amphistegina thrive in water depths between 20-30m. Amphistegina is also commonly associated with sandy sediments in warm, tropical environments with their robust tests able to withstand high energy. Other smaller benthic foraminifera can be found in bathyal environments especially where they are agglutinating and not made of calcite (see CCD below).
Planktonic foraminifera are usually only found in the inner to outer neritic environment, but never within bathyal environments due to the ‘Carbonate Compensation Depth’ (or CCD), a depth below which carbonate is dissolved, typically this boundary is found between 4-5km. Occasionally planktonic foraminifera can be washed in to much shallower environments but these environments should be interpreted based on the greater number of benthic foraminifera than planktonics within the fossil assemblage.

Species of larger benthic and planktonic foraminifera have short temporal ranges which makes them perfect index fossils in determining the age of rocks through biostratigraphy. Good index fossils must fulfill several criteria 1) they must be common and easy to find, 2) they must be cosmopolitan (although migrations must be accounted for) so they can be correlated across the globe, 3) they must be easy to identify so they can be spotted in samples with varying degrees of preservation, 4) they must have short evolutionary histories so that one (preferably several) species can be used to define a particular period of time. Many smaller benthic foraminifera are hardy organisms that have long temporal ranges which make them not particularly useful for biostratigraphic purposes but, as previously illustrated, may have palaeoenvrionmental significance.
I will give more details on the principles of biostratigraphy in a later post.
References:
Langer, M. R., Silk, M. T. B., Lipps, J. H. 1997. Global ocean carbonate and carbon dioxide production: The role of reef Foraminifera, Journal of Foraminiferal Research, 27(4), 271–277
Myers, E.H. 1941. Ecologic relationships of larger foraminifera: Report of the Committee on Marine Ecology as related to Paleontology, Division of Geology and Geography, National Research Council Annual Report, Appendix H, 1945.

